scFv Antibody Construction Service
Single-chain fragment variables (scFv), is a combination of only the variable region of heavy chain (VH) and variable region of light chain (VL) domains. They are mainly constructed by three different methods: scFv generated from synthetic DNA, hybridoma technology or via accession number.
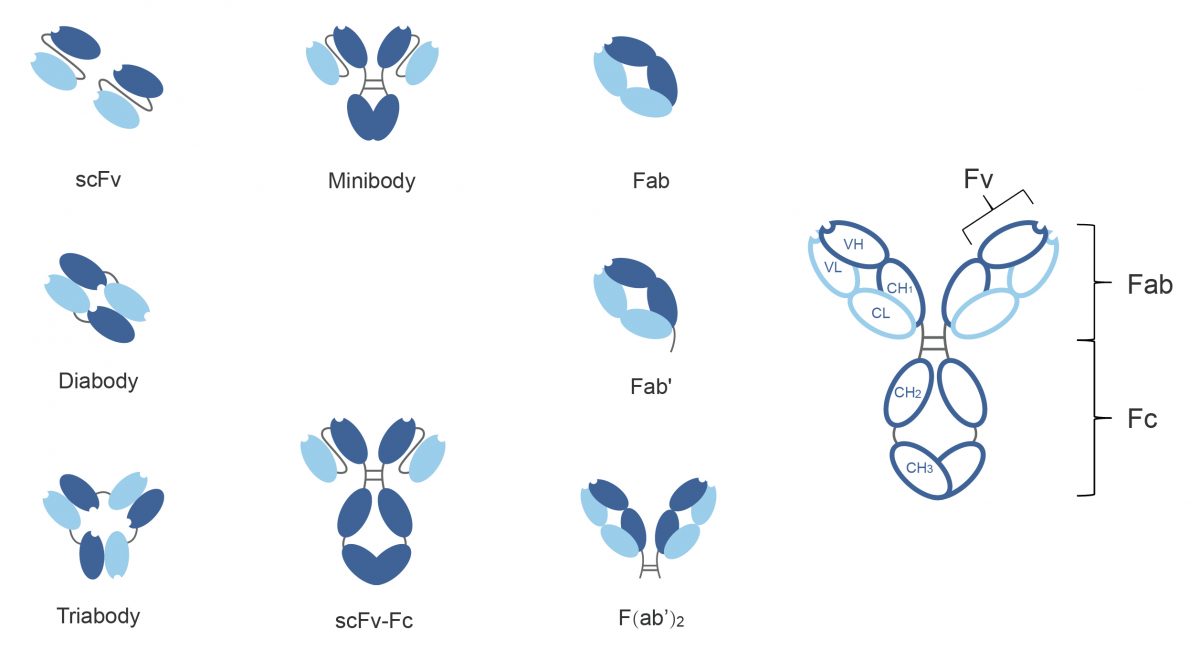
With years of research experience in the field of recombinant antibody construction and expression, Biologics International Corp (BIC) offers antibody fragment production services, which include scFv, diabody, triabody, minibody, scFv-Fc, Fab, Fab’, scFab, and F(ab’)2. Do not hesitate to get in touch for any further information regarding antibody fragments. We are always glad to assist you.
scFv
An scFv is not actually an antibody fragment, but instead is a fusion protein of VH and VL of immunoglobulins, connected with a short linker peptide of 10 to about 25 amino acids. These functional proteins having both antigen-binding capacity and marker activity can be constructed from hybridoma and be applied in the biological applications such as in biosensors, therapeutic gene delivery, and cancer treatment [1]. In addition, the murine antibody can be humanized for its use in therapy, which is rapidly achievable at the scFv stage [2].
scFv Fragments Construction
To obtain scFv fragments, mRNA is first isolated from hybridoma (or spleen cells and B lymphocytes) followed by reverse transcribed into cDNA to serve as a template for the polymerase chain reaction (PCR) amplification of the antibody genes. With this method, large libraries with a diverse range of antibody VH and VL genes could be created [3]. Using the techniques of affinity selection, the scFv fragment with the best affinity and specificity which is displayed on phage coat can be obtained. In the scFv construction, the order of the domains can be either VH-linker-VL or VL-linker-VH [4]. The specific antibodies produced from one selected hybridoma are essentially the same. When the antibody genes were successfully cloned and sequenced, the scFv fragments could be directly expressed in bacterial systems (i.e., E.coli) or mammalian systems (i.e., CHO or HEK293 cells).
scFv Antibody Library
Currently, most recombinant antibody fragments are generated by phage display antibody library [5]. Furthermore, scFv antibody library has several advantages against monoclonal antibodies. First, scFv antibody library makes it possible to select new antibodies which are difficult or impossible to gain through traditional methods, such as human antibodies and antibodies against self-antigens. Second, scFv antibodies with higher affinity could be generated through site-directed mutagenesis, which is easier to be performed. Third, the phages are easily stored or produced. If you need an antibody against some unusual antigens, get in touch, our antibody library with rich diversity and large scale will help you to find an ideal antibody!
More scFv Formats
According to some research, scFv fragments often have lower affinity compared to the parental whole antibody or Fab molecule [6-9], and the scFv fragments complexed into dimers (diabodies), trimers (triabodies) or larger aggregates may be better selections.
The scFv linkers which connect VH and VL domains usually contain 10 to about 25 amino acids. But an scFv molecule with a linker of 5-12 residues long cannot fold into a functional Fv domain and instead associates with a another scFv molecule to form a bivalent dimer (diabody). Further more, if the length of a linker is reduced to one or two glycine residues, the formation of diabodies will be prevented and three scFv fragments will turn to a trimer (triabody) [10].
Other scFv formats such as minibodies, which are composed of a pair of scFv fragments linked via a constant region of heavy chain 3 (CH3) domains, may increase the affinity to antigen and the value to biotechnological applications. Moreover, scFv-Fc fragments have the whole fragment crystallizable (Fc) region which is essential in mediating cytotoxic effector functions. Bispecific diabodies are made by combination of two scFv fragments with different specificities. The VL of an scFv with one specificity is joined via a shortened linker to the VH of an scFv with another specificity and vice versa [10].
Have some antibody formats in mind? Contact us with your ideas, let's see how can assist you.
scFv vs. Fab Fragment
| Fragment Type | scFv | Fab |
|---|---|---|
| Composition | VH, VL | VH, VL,CH1, CL |
| M.W. (kDa) | 30 | 50 |
| Blood clearance | Very rapid | Rapid |
| Tumor penetration | Great | Good |
| Retention times | Very short | Short |
| Affinity | Reduced | Better than scFv |
| Stability | Unstable over longer periods | High stability in long-term storage |
| Production |
|
|
References
- Victor Crivianu-Gaita, Michael Thompson. Aptamers, antibody scFv, and antibody Fab fragments: an overview and comparison of three of the most versatile biosensor biorecognition elements. Biosensors and Bioelectronics, vol.85, 2016, pp.32-45.
- Jonas V. Schaefer, Annemarie Honegger, et al. Construction of scFv fragments from hybridoma or spleen cells by PCR assembly. Antibody Engineering, Chapter 3, pp.21-44.
- James D.Marks, Hennie R.Hoogenboom, et al. By-passing immunization: human antibodies from V-gene libraries displayed on phage. Molecular Biology, vol.222, 1991, pp.581-597.
- Zuhaida Asra Ahmad, Swee Keong Yeap, et al. scFv antibody: principles and clinical application. Clinical and Developmental Immunology, vol.2012, 2012, pp.1-15.
- Michael Hust, Thomas Jostock, et al. Single chain Fab (scFab) fragment. BMC Biotechnology, 2007, 7:14.
- Hennie R.Hoogenboom, Andrew D.Griffiths, et al. Multi-subunit proteins on the surface of filamentous phage: methodologies for displaying antibody (Fab) heavy and light chains. Nucleic Acids Research, vol.19, 1991, pp.4133-4137.
- Carl A K Borrebaeck, A C Malmborg, et al. Kinetic analysis of recombinant antibody-antigen interactions: relation between structural domains and antigen binding. Biotechnology (N Y), vol.10, 1992, pp.697-698.
- Glockshuber R, Malia M, et al. A comparison of strategies to stabilize immunoglobulin Fv-fragments. Bio-chemistry, vol.29, 1990, pp.1362-1367.
- William D. Mallender, Jenny Carrero, et al. Comparative properties of the single chain antibody and Fv derivatives of mAb 4-4-20. Relationship between interdomain interactions and the high affinity for fluorescein ligand. The Journal of Biological Chemistry, vol.271, 1996, pp.5338-5346.
- John L.Atwell, Kerry A.Brehency, et al. scFv multimers of the anti-neuraminidase antibody NC10: length of the linker between VH and VL domains dictates precisely the transition between diabodies and triabodies. Protein Engineering, vol.12, 1999, pp.597-604.
Contact Us


